Building a Test Evidence Strategy That Scales
Learn how to build a scalable test evidence strategy. Capture screenshots, logs, and artifacts consistently to pass audits and debug faster.
Insights, tutorials, and updates on software testing and QA

Learn how to build a scalable test evidence strategy. Capture screenshots, logs, and artifacts consistently to pass audits and debug faster.

Vibe coding is fast until your AI starts hallucinating. Learn how connecting your test management tool via MCP creates guardrails that keep AI grounded in reality.

Manage test cases directly from your AI coding assistant with TestCollab MCP Server. Create, update, and organize tests without leaving your development environment.

AI has made code creation cheap, but verification remains hard. Learn why testing is now about defining specifications upfront, not catching bugs at the end.

Create custom execution statuses in TestCollab. Learn setup, priority rules, and best practices to improve QA reporting.

Learn how our team improved regression testing using TestCollab, an AI-powered test management tool. Discover practical strategies, common pitfalls, and tips to build a stable, scalable QA process with automation and test data management.

Prepare your QA strategy for the European Accessibility Act 2025: learn how integrated test management streamlines accessibility testing & compliance.

Boost coverage without duplication—TestCollab’s new Test Datasets & Parameters enable fast, data-driven test management with reusable test data and dynamic parameters.

Discover the key differences between verification and validation in software testing. Learn why both are crucial for quality assurance and how modern test management tools like TestCollab can streamline the process.

Launching QA Copilot CI—plug-and-play ai testing automation for GitHub, GitLab & Jenkins. Auto-healing tests, smart retries, video evidence, user-driven roadmap.

TestCollab is retiring its Free Plan on August 4, 2025. Secure a 90-day grace period for your test management projects—upgrade to Premium now to keep your data intact.
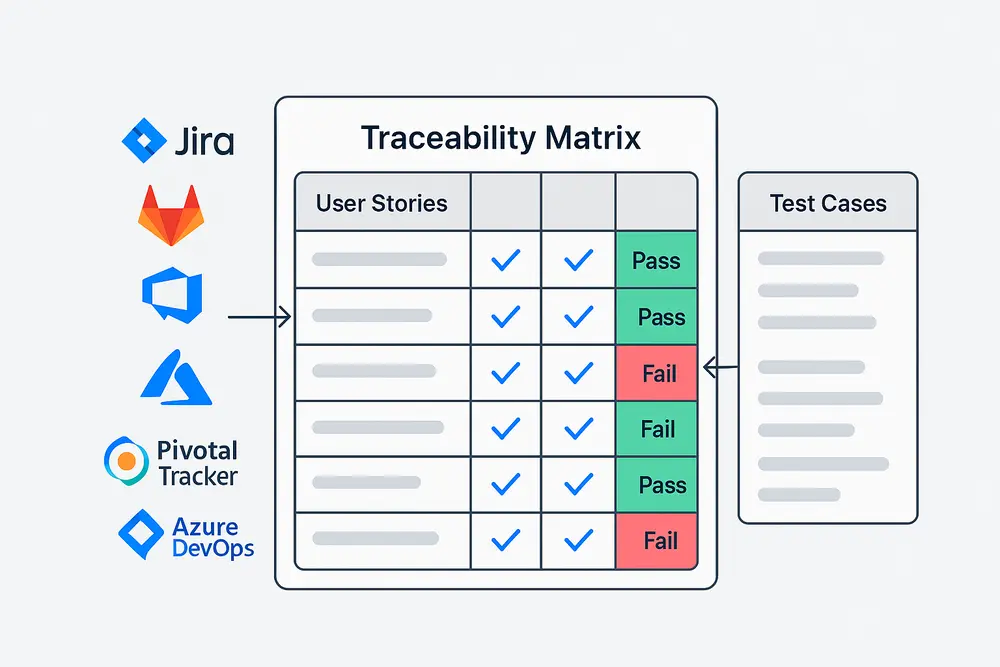
Struggling with gaps in requirement traceability? Discover how TestCollab’s test management tool gives QA teams end-to-end visibility, dynamic requirement traceability matrix, and integrations with Jira, GitLab, and more—without the spreadsheet mess.